Cell and gene therapies (CGT) changed our approach in how we treat human disease. Unlike traditional drugs that often manage symptoms, CGTs offer durable, and sometimes curative, solutions by correcting the underlying cause of a disease. Specifically speaking to cell therapies, they are not like traditional drug modalities. They are living therapies engineered to restore biological function or grant new abilities to a patient’s own cells, targeting everything from inherited disorders like sickle-cell anemia to complex cancers.
The term CGT encompasses a broad class of treatments. In its purest form, cell therapy involves treating disease by transferring cells with a desired function into the patient’s body therefore making the therapy product, the cell itself. These therapeutic cells can be sourced from:
- The patient themselves (autologous)
- A matched donor (allogeneic)
- An established, well-characterized cell line
Gene therapy, on the other hand, involves the transfer of DNA or the direct alteration of a patient’s genome to achieve a therapeutic effect. Many modern treatments are a hybrid of both. Chimeric antigen receptor (CAR) T-cell therapy, for example, is a prime illustration. It involves taking a patient’s immune cells (T-cells), genetically engineering them to express receptors that can recognize and attack cancer. These CAR-T-cell therapies are then re-infused into the body of the patient for treatment.
CGTs are delivered using one of two major strategies: ex vivo or in vivo.
- Ex vivo (outside the body): Cells are removed from the patient, modified in a controlled manufacturing environment, and then administered back to the patient as a final product. This allows for extensive quality control before the treatment reaches the patient.
- In vivo (inside the body): A gene therapy vector or editing tool is delivered directly into the patient, where the genetic modification occurs. This approach turns the patient’s body into the “manufacturing” site, which presents unique challenges for delivery, targeting, and safety assessment.
The choice between ex vivo and in vivo is a critical decision that dictates the entire development, manufacturing, and regulatory pathway for a new cell and gene therapy.
To engineer a cell or gene therapy, most cases require the delivery of a genetic payload. The delivery of genetic payloads is often accomplished using viral vectors like lentiviruses and adeno-associated viruses (AAVs), which are highly efficient at entering cells. Non-viral systems, such as lipid nanoparticles (LNPs), are another delivery approach and are quite favorable because of their manufacturing scalability and ability to deliver RNA-based medicines without altering the host genome.
The Importance of Single-Cell Multiomic Analytical Assays
Cell and gene therapy analytical development is the foundation of evaluating the therapy’s safety, toxicity, potency, and efficacy. Unlike a known small chemical molecule drug, a cell therapy product is a population of living, genetically modified cells with significant variability. The therapy is the cell, making its characterization essential before being administered into patients.
Conventional “bulk” analytical assays, which average signals from millions of cells, can hide rare but critical events like off-target genetic edits, inconsistent vector delivery, or the growth of unwanted cell clones. These hidden subpopulations can reduce the therapy’s effectiveness or even cause harm. Furthermore, this traditional approach is laborious, as it requires developers to perform multiple, separate assays to measure different attributes such as editing efficiency, gene expression readouts, or phenotypic states and then attempt to combine the fragmented results just to try and get a full understanding of their therapy (Fig 1a).
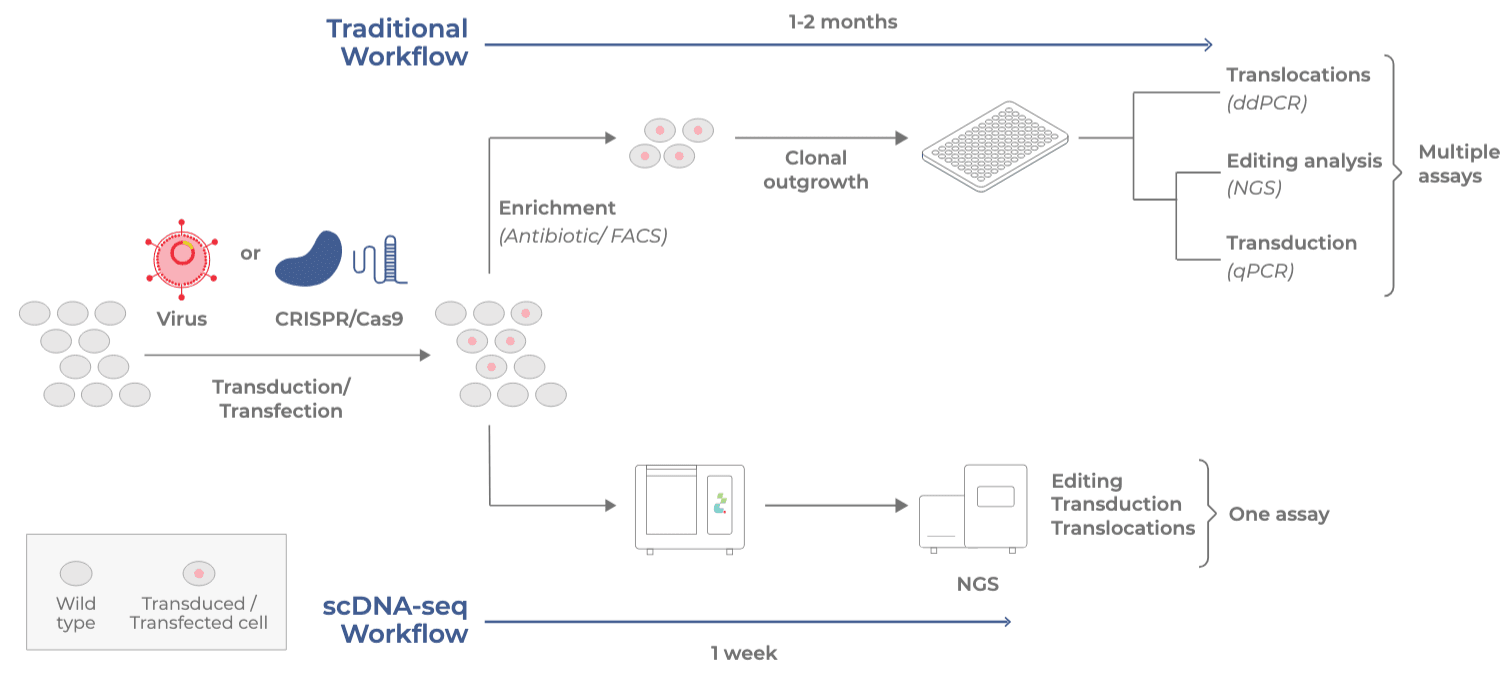
To overcome this, developers rely on custom, single-cell multiomic analytical development assays (Fig 1b). These powerful GxP deployable single-cell multiomic assays provide the high resolution needed to measure key quality attributes in thousands of individual cells at once (Fig 2). This includes but not limited to:
- Vector Copy Number (VCN): Confirming how many copies of the therapeutic gene are in each cell.
- On/Off-Target Editing: Verifying that genetic edits occurred at the correct location without unintended changes elsewhere.
- Zygosity and Integration: Determining if the vector has integrated into the chromosomes and assessing the nature of the edit and co-occurance.
- Genome Integrity: Ensuring the editing process did not cause large-scale damage to the DNA or cause chromosomal aberrations.
- Translocations: a type of rearrangement in which sections of DNA are exchanged within or across chromosomes.

By linking a cell’s genetic makeup (genotype) to its RNA expression, which directly governs the cell’s functional state (phenotype), these custom, single-cell multiomic assays provide a deep understanding of a cell and gene therapy to establish its potency and safety leading to its success in patients.
Accurate characterization of cell and gene therapy product requires resolving cellular heterogeneity that is obscured by bulk product averages. Single-cell multiomics facilitates the simultaneous quantification of genomic integrity and proteomic expression, allowing for the direct correlation of genotype to phenotype at the individual cell level. This resolution is fundamental to the development of custom fit-for-purpose assays designed to monitor Critical Quality Attributes (CQAs) such as vector copy number distribution and off-target editing rates. The following “Frequently Asked Questions” address specific technical challenges in analytical development, detailing how single-cell methodologies are applied to validate product safety, potency, and purity needed for regulatory reporting.
Frequently Asked Questions
How do we prove purity and identity of the final cell therapy product?
Using a custom cell and gene therapy assay that leverages single-cell multiomics, you can resolve rare contaminating cell types or identify unwanted off-targets at the individual cell level that bulk assays cannot. These custom cell and gene therapy assays provide deep confirmation of your final cell therapy product by combining genome evaluation with gene expression, surface marker identification, and clonal lineage tracking at single-cell resolution.
How should we measure vector copy number (VCN) and transduction efficiency?
Conventional methods for measuring vector copy number (VCN) and transduction efficiency rely on combining bulk technologies such as flow cytometry with qPCR or ddPCR. Flow cytometry provides the fraction of marker-positive cells, while qPCR or ddPCR reports only the average VCN across the population. These stitched-together insights cannot show how copies are distributed across individual cells, which means rare high-VCN clones or large non-transduced subpopulations can be completely missed. Single-cell assays overcome these limitations by quantifying VCN in thousands of individual cells, revealing the true dose distribution and providing a direct, cell-level measure of transduction efficiency. This single-cell approach delivers a more accurate and clinically relevant assessment of product safety, potency, and consistency than any bulk measurement strategy.
How do we assess off-target editing and genomic safety for gene editing therapies?
We assess genomic safety by simultaneously measuring both the intended on-target edits and any unintended off-target edits within the same single cell. This approach provides the clearest view of the risk-benefit profile by directly answering the critical question: “Are the cells with potentially dangerous off-target edits the same ones that contain the desired therapeutic modification?”
How do we measure immunogenicity?
We use single-cell multiomics to resolve the clonal architecture of the immune response by simultaneously sequencing TCR/BCR loci and quantifying surface protein expression. This directly links antigen-specific clonotypes to functional phenotypes, revealing whether vector-reactive T cells form short-term responses or establish long-term memory populations.
Why should I consider single-cell multiomic methods for cell and gene therapy assay development?
Bulk cell and gene therapy assays average results across a population and can mask rare but dangerous changes within the therapy product. Single-cell multiomics measures DNA, RNA, and protein in individual cells, revealing hidden risks like off-target edits, high vector copy numbers, or clonal dominance that directly affect safety and efficacy.
How can single-cell assays de-risk my cell and gene therapy program?
Every cell is a potential liability because in some cases, it is the therapy product. With custom cell and gene therapy assays that leverage single-cell multiomics, you can detect rare but critical safety issues such as chromosomal aberrations, unintended edits, or aberrant vector integrations before they derail clinical development. This helps avoid costly clinical milestones and strengthens regulatory submissions.
What types of attributes can be measured with single-cell multiomics in cell & gene therapy development?
Custom cell and gene therapy assays that leverage single-cell multiomics should measure vector copy number (VCN), multiplex edits, chromosomal integrity, zygosity, on/off-target editing, immunophenotyping, and targeted gene expression all within the same cell, providing a complete profile of therapeutic quality, safety, and efficacy.
Why are analytical development strategies so critical in cell & gene therapy?
Unlike molecular drugs, the therapeutic product in cell and gene therapies can be the cell itself, a living heterogeneous population. Without rigorous analytical development, hidden variability threatens potency, persistence, and safety. Single-cell analytical strategies ensure rare but consequential events are identified and controlled.
How do single-cell assays strengthen potency assay development?
Single-cell data directly links genetic modifications to functional readouts (e.g., gene expression changes or cell surface phenotype). This enables developers to pinpoint the exact subpopulations driving efficacy and build potency assays that truly understand therapeutic mechanisms of action.
What role do single-cell assays play in monitoring safety events like off-target editing?
Bulk cell and gene therapy assays may show “clean” averages, but single-cell assays reveal whether the same cells with therapeutic edits also carry unintended off-targets or structural variants. This adds a layer of genomic safety monitoring that regulators are increasingly prioritizing.
How does assay transfer work for cell & gene therapy manufacturing and scale-up?
Mission Bio’s Cell and Gene Therapy Assay Development Services include full assay transfer to your site or CRO/CDMO, with Tapestri instrument installation, IOQ/PQ documentation, SOPs, bioinformatics support, and training. This ensures your custom assays are GMP-ready, reproducible, and consistent across all phases of therapy development.
Can single-cell assays help demonstrate comparability after a manufacturing change?
Yes. Single-cell multiomics analysis from our custom cell and gene therapy assays reveals whether cellular composition, clonal diversity, and functional states remain consistent. This provides deeper evidence than bulk assays alone, supporting regulatory comparability requirements and de-risking manufacturing transitions.




